User Tools
Site Tools
Site Tools
Table of Contents
Datacenter cooling
This page regroup important basis related to cooling in general. This is the result of my own experience and I hope it will help you.
Water
General tips
PH
PH is an excellent an cheap to measure indicator of water health. It should be between 7 and 9.
If PH suddenly drop (< 6) and smell like formic acid, check presence of glycol in the circuit !
When exposed to aluminum/coper and to air in a circuit, glycol mixed with water can generate acide, causing a student PH drop. Many related paper can be found on the National Institute of Standards and Technology web site : http://fire.nist.gov
http://www.veoliawaterstna.com/crownsolutions/ressources/documents/2/21823,Glycol.pdf
“Aqueous solutions of ethylene glycol and propylene glycol produce acidic degradation products upon thermal oxidation. This results in a decrease in pH of the aqueous solutions which could accelerate the corrosion of metallic components.”
Galvanic corrosion
THIS thing is the worst nightmare of datacenter cooling manager.
“Galvanic corrosion is an electrochemical process in which one metal corrodes preferentially to another when both metals are in electrical contact, in the presence of an electrolyte. This same galvanic reaction is exploited in primary batteries to generate an electrical voltage.” (Wikipedia)
Most datacenter are affected by galvanic corrosion, especially when different materials like copper and aluminum are used in the hydraulic systems. This generates surface deterioration and particles in the flow. Galvanic corrosion is a very well know effect in amateur water-cooling communities.
·
Galvanic corrosion in waterblocks (http://www.overclock.net/t/1200435/my-apogee-gtx-fail-epic-example-of-galvanic-corrosion, pn0yb0i)
·
·
Galvanic corrosion in waterblocks (https://linustechtips.com/main/topic/193492-mixing-fitting-brands/, Vacsol)
·
When mixing two metals with an electrolyte (water), you create a cell. The more “anodic” (or “less noble”) metal will be the negative terminal (anode) of the cell thus made, and will oxidize.
To evaluate the intensity of the effect on a site, list all materials and compare their anodic index (this parameter is a measure of the electrochemical voltage that will be developed between the metal and gold).
| Metal | Index (V) |
|---|---|
| Most Cathodic | |
| Gold, solid and plated, Gold-platinum alloy | -0.00 |
| Rhodium plated on silver-plated copper | -0.05 |
| Silver, solid or plated; monel metal. High nickel-copper alloys | -0.15 |
| Nickel, solid or plated, titanium an s alloys, Monel | -0.30 |
| Copper, solid or plated; low brasses or bronzes; silver solder; German silvery high copper-nickel alloys; nickel-chromium alloys | -0.35 |
| Brass and bronzes | -0.40 |
| High brasses and bronzes | -0.45 |
| 18% chromium type corrosion-resistant steels | -0.50 |
| Chromium plated; tin plated; 12% chromium type corrosion-resistant steels | -0.60 |
| Tin-plate; tin-lead solder | -0.65 |
| Lead, solid or plated; high lead alloys | -0.70 |
| 2000 series wrought aluminum | -0.75 |
| Iron, wrought, gray or malleable, plain carbon and low alloy steels | -0.85 |
| Aluminum, wrought alloys other than 2000 series aluminum, cast alloys of the silicon type | -0.90 |
| Aluminum, cast alloys other than silicon type, cadmium, plated and chromate | -0.95 |
| Hot-dip-zinc plate; galvanized steel | -1.20 |
| Zinc, wrought; zinc-base die-casting alloys; zinc plated | -1.25 |
| Magnesium & magnesium-base alloys, cast or wrought | -1.75 |
| Beryllium | -1.85 |
| Most Anodic | |
In most datacenter, heat exchangers are in aluminum and many valves/other hydraulic components are in copper or brass (copper + zinc) (especially inside chillers and air handling unit), creating galvanic effect.
Presence of particles in the water (or abnormal color), pressure rising in pipes, or voltage between pipes and ground, are mainly due to galvanic corrosion.
Air
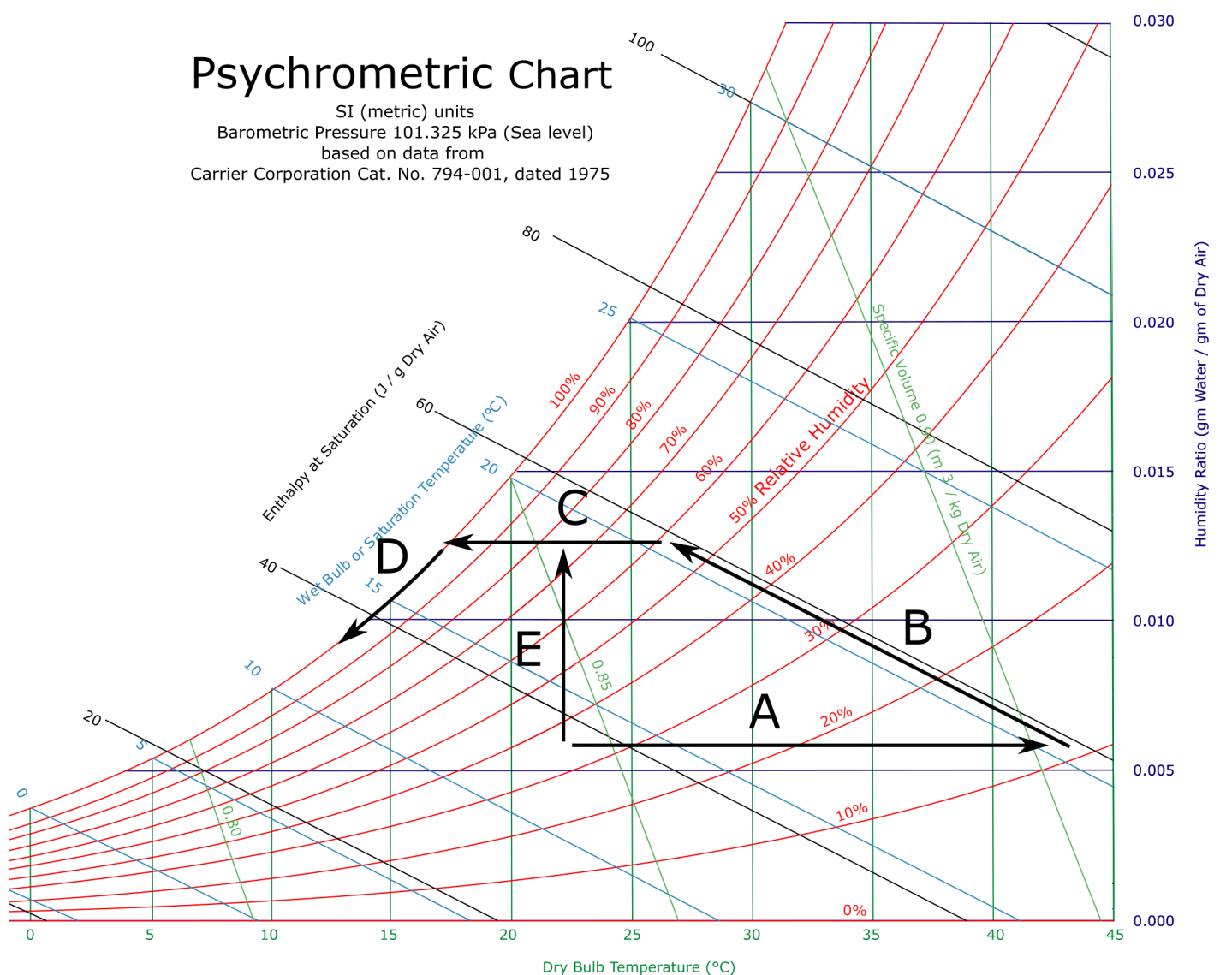
Psychometric and vaporization charts
Humid air is mainly used for room temperature and humidity management, but also for outside air with adiabatic coolers. Cooling manager must know how to use the psychometric chart to detect energy losses or possible water condensing.
- A. Air heating. (Example: IT equipment, heating, dry cooler, etc.)
- B. Adiabatic humidification, using cold water. (Example: adiabatic coolers, etc.)
- C. Air cooling, without water. (Example: air handling units, etc.)
- D. Air cooling with saturated humidity. (Example: near heat exchangers inside air handling units or inside cooled doors, etc.)
- E. Air humidification, using vapor. (Example: humidifier, etc.)
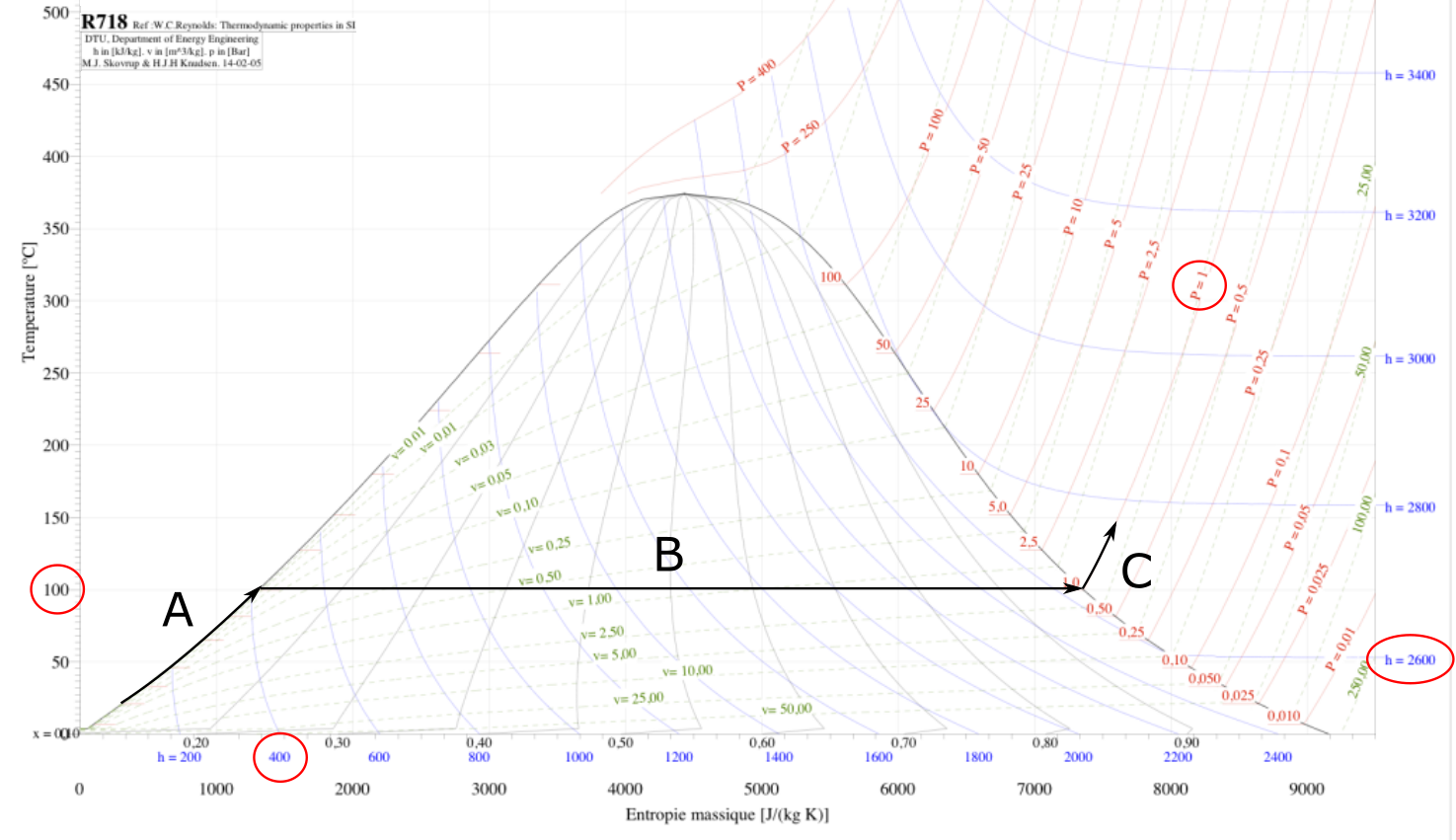
It is also interesting to understand vaporization chart, which highlight how much energy is needed/evacuated to change phase.
Note: values are only given for example.
- A. Heating water from cold water to 100°c at P = 1 atm.
- B. Phase change, from liquid water to vapor. Temperature is fixed at 100°c, but enthalpy rise from 400 J/(Kg.K) to more than 2600 J/(Kg.K).
- C. Vapor heating, from 100°c to near 150°c, keeping 1 atm pressure but rising enthalpy.
Formulae
COP
Cooler performance is defined by the COP (coefficients-of-performance). COP is defined by:
COP = (Cooling power) / (Input electrical power)
Bernoulli
For liquids and low mach gas flows, it is possible to assume fluids to be incompressible, and use Bernoulli principle :
<m 18>{v^2/{2g}}+z+{p/{rho g}}=constant</m>
With :
- <m 18>v</m> the fluid flow speed at a point on a streamline,
- <m 18>g</m> the value of acceleration due to gravity,
- <m 18>z</m> the elevation of the point above a reference plane, with the positive z-direction pointing upward – so in the direction opposite to the gravitational acceleration,
- <m 18>p</m> the pressure at the chosen point, and
- <m 18>rho</m> the density of the fluid at all points in the fluid.
Between two points (1 and 2) of a flow (in a pipe for example), it is possible to assume with no pressure losses :
<m 18>2g+z_1+{p_1/{rho g}}=2g+z_2+{p_2/{rho g}}</m>
Now considering pressure losses :
<m 18>2g+z_1+{p_1/{rho g}} + H_1 =2g+z_2+{p_2/{rho g}} + H_2</m>
Pressure losses
There are two types of pressure losses :
- linear losses, friction of fluids along a pipe or a surface on a defined length
- singular losses, due to singular flow perturbation, for example a bent pipe or an exhaust.
Of course, losses are logical : curved shapes generates less losses than angular shapes. Same with pipes, the bigger the pipe, the less losses.
A good example is the following table, providing coefficients for singular losses, have a look on differences between shapes :
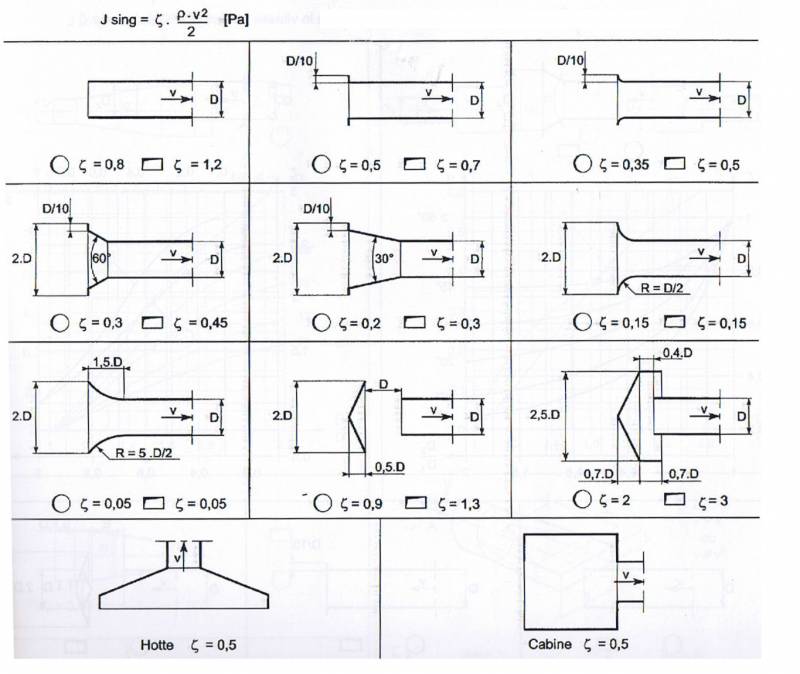 (from http://www.mecaflux.fr/perte%20charge%20singuliere.html)
(from http://www.mecaflux.fr/perte%20charge%20singuliere.html)